Research Hub

How to Read a Certificate of Analysis (COA) | WhyNot Labs Research Hub
How to Read a Certificate of Analysis (COA): A Visual Guide for Researchers
What each section means, red flags to watch for, and how to verify research peptide quality before it enters your lab.
⚠️ FOR RESEARCH USE ONLY. All compounds referenced in this article are intended exclusively for laboratory research purposes. Not for human or animal consumption. These products have not been evaluated by the FDA.
A Certificate of Analysis (COA) is an independent laboratory document that verifies a research compound's identity, purity, and quantity. It's the single most important quality indicator when purchasing research peptides. Here's how to read one and what each section actually means.
COA at a Glance
| COA Section | What It Tells You | What to Look For |
|---|---|---|
| Lab Identification | Who performed the testing | Third-party lab name, address, accreditation |
| Sample Identification | What was tested | Batch number, compound name, date received |
| Identity Verification | Is this the right compound? | Mass spectrometry confirmation |
| Purity Analysis | How pure is it? | HPLC results, ≥98% for research-grade |
| Quantity Verification | Does the label match? | Actual mg vs. stated mg on the label |
| Testing Methodology | How was it tested? | HPLC, Mass Spec, or both |
| Date of Analysis | When was it tested? | Recent date, ideally within weeks of sale |
Why COAs Matter (And Why Most Companies Don't Publish Them)
Here's the thing. A COA is the only objective proof that a research compound is what it claims to be. Without one, you're trusting a label. That's it. A label somebody printed.
So why don't most peptide vendors publish their COAs?
Good question. Some don't test at all. Others do in-house testing, which is basically grading your own homework. And a few test with independent labs but quietly keep the results hidden, making you email support and wait three days for a PDF that may or may not match your batch.
If a company can't show you a third-party COA for the specific batch you're buying, that tells you everything you need to know.
And that's the real issue. Anyone can throw a label on a vial. A real COA from a real lab? That's not something you can fake. (Well, some try. We'll cover how to spot fakes below.)
All research compounds should be verified through independent analysis before use in any laboratory setting. COAs are the industry standard for this verification.
Anatomy of a COA
A proper Certificate of Analysis isn't complicated once you know what you're looking at. Each section serves a specific purpose.
1. Lab Identification
The "who tested it" section. It sits at the top of the document and should include the laboratory's name, physical address, and contact information. Some labs also include accreditation numbers or certification logos.
The critical distinction: third-party vs. in-house. A third-party lab has zero financial incentive to fudge results. They don't care if the sample passes or fails. An in-house test means the company selling you the product is also the one telling you it's good.
2. Sample Identification
This section confirms exactly what was submitted for testing. You'll see the compound name, the batch or lot number, and usually the date the sample was received by the lab.
The batch number on the COA needs to match the batch number on your product. If it doesn't, the COA is irrelevant to what you're holding. A company could test one batch in 2023 and use that same COA for every shipment through 2026.
3. Identity Verification
This is the "is this actually what it says it is?" test. The lab uses mass spectrometry to confirm the molecular identity of the compound.
Mass spectrometry works by measuring a compound's molecular weight — essentially a molecular fingerprint. If a compound is labeled as BPC-157 (molecular weight 1419.53 g/mol, CAS: 137525-51-0), the mass spec results need to confirm that exact molecular weight.
This is a pass/fail section. Either the compound is what it claims to be, or it isn't. No gray area.
4. Purity Analysis Most Important
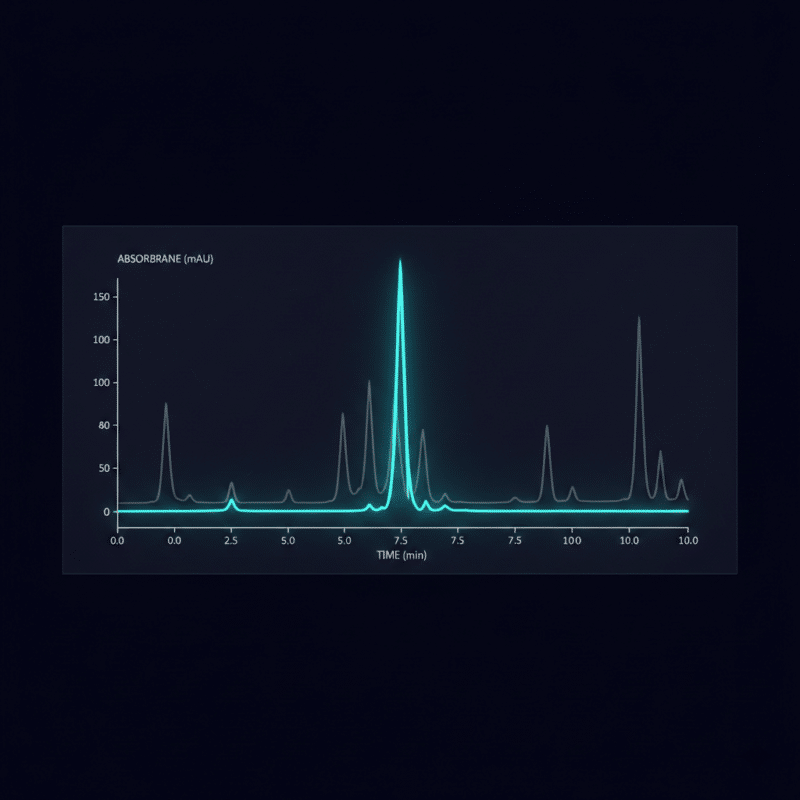
Purity analysis, typically run through HPLC (High-Performance Liquid Chromatography), tells you what percentage of the sample is the actual target compound versus impurities.
Research-grade peptides should show ≥98% purity. That's the baseline. Anything below 95% raises serious questions. Above 99%? That's excellent.
Most buyers miss this: the purity percentage alone isn't the full story. Look for the actual HPLC chromatogram (the graph) if possible. A clean chromatogram shows one dominant peak with minimal noise. Multiple large peaks mean impurities or degradation.
5. Quantity Verification
Does the vial actually contain what the label says? If a product is labeled "30mg," this section confirms whether the actual content matches.
Underfilling is a real issue in the peptide market. A vial labeled 10mg might contain 7mg or 8mg. Without quantity verification, you'd never know. Look for measured quantity vs. stated quantity. Perfect matches are rare in analytical chemistry (don't expect exactly 10.000mg), but if a 10mg vial tests at 6.2mg, that vendor has some explaining to do.
6. Testing Methodology
This section lists the analytical methods used. You'll typically see HPLC and Mass Spectrometry (MS), sometimes listed as LC-MS when combined.
HPLC
Separates components and measures how much of each is present. Tells you purity. Think of it as sorting and counting everything in the vial.
Mass Spectrometry
Measures molecular weight of compounds. Confirms identity. HPLC alone can't tell you it's the right compound. You need both.
7. Date of Analysis
When was this test actually performed? Recent is better. Peptides can degrade over time, and a COA from two years ago tells you nothing about the compound in your package today. You want testing that's current and tied to the batch you're purchasing.
COAs provide essential quality verification for research compounds. Researchers should always verify independent testing documentation before incorporating any compound into laboratory protocols.
Red Flags on a COA: What Should Make You Suspicious
Not all COAs are created equal. Some aren't even real. Here's what should trigger your skepticism.
⚠ No lab name or address
A legitimate COA comes from a legitimate lab. If there's no identifiable laboratory, no address, and no way to verify the testing facility exists, treat the document as worthless.
⚠ In-house testing only
In-house testing means the company tested its own product. Would you trust a restaurant health inspection performed by the restaurant's owner? Same logic applies here.
⚠ Missing methodology details
A real COA specifies how the testing was performed. If it just says "tested" with a purity number and nothing else, that's a template someone filled out, not an analytical report.
⚠ Batch numbers that don't match your product
Easiest check you can do. Batch number on the COA vs. batch number on your vial or packaging. If they don't match, that COA isn't for your product.
⚠ Suspiciously round numbers
Analytical chemistry doesn't produce perfectly round results. A purity reading of exactly 99.00% or a quantity of exactly 10.000mg should raise eyebrows. Real lab results have decimal variation — something like 98.73% purity or 10.14mg content looks legitimate. Perfect round numbers look fabricated.
⚠ Outdated or missing dates
No date at all is a red flag. A date from years ago paired with a product you bought this week is another. A COA dated before the company even launched? That happens more often than you'd expect.
How WhyNot Labs Handles COAs
We don't ask you to trust us. We show you the proof.
Every batch we sell gets shipped to Vanguard Laboratory before a single vial goes out the door. Not some batches. Not the first batch then skip the rest. Every batch.
| Step | What Happens |
|---|---|
| 1 | We source a compound from our verified manufacturers |
| 2 | Before anything ships, a sample goes to Vanguard Laboratory |
| 3 | Vanguard runs full independent analysis: identity, purity, and quantity verification |
| 4 | If it passes: COA published, shipping begins. If it fails: entire batch rejected. Period. |
| 5 | Full COA published in our COA Library — publicly, for anyone to view |
And we don't just post them on a hidden page somewhere. Every order ships with a COA card featuring a QR code that links directly to the specific certificate for that batch. Scan it, read it, verify it yourself.
What Vanguard Laboratory Tests For
Vanguard Laboratory is an independent third-party laboratory. They don't work for us. They don't have a financial relationship with us beyond the testing fee. They don't care if our products pass or fail.
That independence is the entire point.
Identity
Confirms the compound is actually what it's labeled as, using mass spectrometry to match molecular weight against known reference standards.
Purity
HPLC analysis to determine the percentage of active compound. Our standard is ≥98% purity, though most batches come back higher.
Quantity
Verifies that the milligram content matches what's stated on the label. You paid for a specific amount — we verify you're getting it.
How to Verify Your WhyNot Labs Product
Got your order? Here's how to check it in about 15 seconds.
1. Find the QR code on the COA card included in your box.
2. Open your phone camera and scan it. It takes you directly to your batch's Certificate of Analysis on our website.
3. Check the batch number on the COA against the batch number on your vial label. They should match.
That's it. No emailing support. No waiting. No digging through a website. Scan, verify, done.
You can also visit our COA Library anytime and search by batch number or product name. Every COA we've ever published is there. Publicly.
All WhyNot Labs products undergo independent third-party analysis. COAs are published for every batch and accessible through our COA Library and via QR code on product packaging. For research use only.
At WhyNot Labs, every batch is independently tested by Vanguard Laboratory. Full COAs are published on site and linked to your specific batch number. We source directly from verified manufacturers — not wholesale distributors — because that's the only way to control quality from start to finish.
WhyNot Labs sells research compounds only. Not for human consumption.
Frequently Asked Questions
What is HPLC?
HPLC stands for High-Performance Liquid Chromatography. It's an analytical technique that separates and quantifies the components in a liquid sample. In peptide testing, it determines purity by measuring what percentage of the sample is the target compound versus impurities. Think of it as a molecular sorting machine that counts everything in the vial.
What is mass spectrometry?
Mass spectrometry measures the mass-to-charge ratio of molecules in a sample — in simpler terms, it weighs the molecules. Since every compound has a unique molecular weight, mass spectrometry confirms whether a sample actually contains the compound stated on the label. It's the gold standard for identity verification.
What purity level is acceptable for research-grade peptides?
Research-grade peptides should have ≥98% purity as verified by HPLC. Many quality suppliers consistently exceed 99%. Anything below 95% should be questioned. WhyNot Labs holds all products to a minimum ≥98% purity standard through Vanguard Laboratory testing.
Should I trust in-house COAs?
Short answer? No. In-house testing means the company selling the product is also verifying its own quality. There's an inherent conflict of interest. It's not that in-house labs can't produce accurate results — it's that you have no way to verify their independence or methodology. Always look for third-party testing from a recognized independent laboratory.
How do I verify a COA is real?
Check whether the lab listed on the COA actually exists — do they have a website, a physical address, a reputation? Next, confirm the batch number matches your product. Then examine the details: are testing methods specified? Do results have realistic (non-round) numbers? Are dates current? These checks take about two minutes and reveal most fraudulent documents immediately.
How often should suppliers test their products?
Every batch. Not once a year. Not when they launch a new product. Every single batch that ships should have its own COA from an independent lab. Compounds can vary between production runs, and the only way to know what's in your specific batch is to test your specific batch.
What's the difference between a COA and a spec sheet?
A spec sheet lists intended specifications — what a product should be. A COA shows actual test results for a specific batch — what it actually is. Spec sheets are promises. COAs are receipts. You want the receipt.
Research Use Disclaimer
All products and compounds discussed in this article are intended exclusively for laboratory research purposes. Not for human or animal consumption. These products are not drugs, foods, or cosmetics and have not been evaluated by the FDA. WhyNot Labs makes no claims regarding therapeutic benefits, efficacy, or safety for any purpose.
Written by Ash, Founder of WhyNot Labs. All WhyNot Labs products are independently tested by Vanguard Laboratory with full Certificates of Analysis published for every batch.
Bestseller Products
In stock
Bulk Pricing
| 1 – 4 | Full Price |
| 5 – 9 | 10% OFF |
| 10+ | 15% OFF |
In stock
Bulk Pricing
| 1 – 4 | Full Price |
| 5 – 9 | 10% OFF |
| 10+ | 15% OFF |
In stock
Bulk Pricing
| 1 – 4 | Full Price |
| 5 – 9 | 10% OFF |
| 10+ | 15% OFF |
In stock
Bulk Pricing
| 1 – 4 | Full Price |
| 5 – 9 | 10% OFF |
| 10+ | 15% OFF |